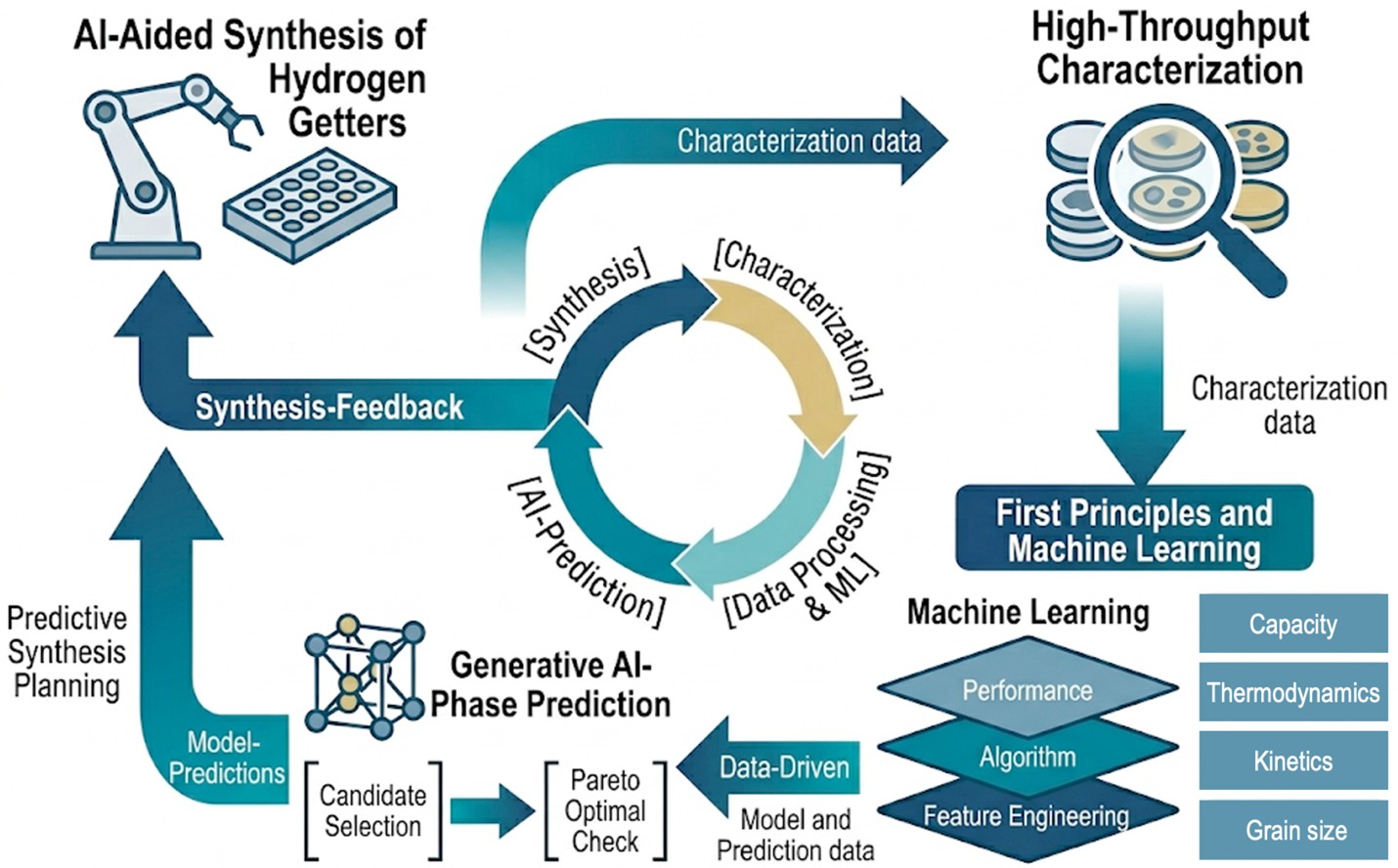
Artificial intelligence and data science present a roadmap to accelerate the discovery of new materials, with important implications in energy generation, storage, and utilization. We are developing explainable AI-driven approaches to systematically tune the properties of high-entropy alloys (HEAs) and high-entropy intermetallics (HEIs) for hydrogen storage, separation, and processing. We determined that feature importance defined by a gradient boosting tree regressor identifies a strong correlation between the equilibrium plateau pressure and an atomic volume descriptor extracted just from the elemental composition of the alloy. These predictions are being incorporated with automated alloy synthesis to realize the vision of a self-driving autonomous lab.
Further Reading:
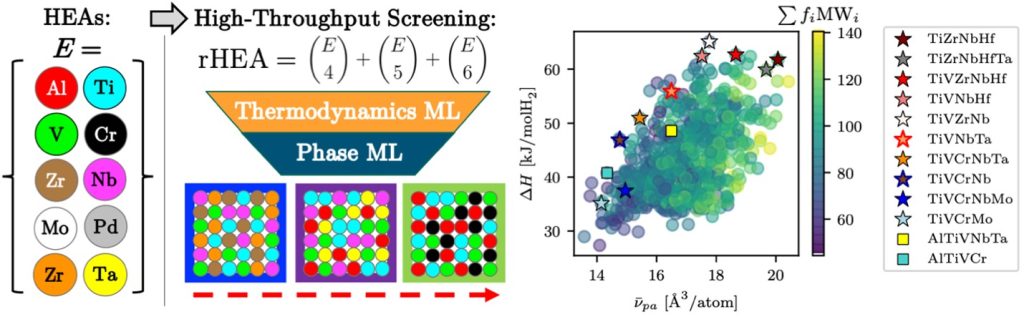
- Witman, M. D.; Ling, S. L.; Wadge, M.; Bouzidi, A.; Pineda-Romero, N.; Clulow, R.; Ek, G.; Chames, J. M.; Allendorf, E. J.; Agarwal, S.; Allendorf, M. D.; Walker, G. S.; Grant, D. M.; Sahlberg, M.; Zlotea, C.; Stavila, V. Towards Pareto optimal high entropy hydrides via data-driven materials discovery. Journal of Materials Chemistry A 2023, 11 (29), 15878-15888. DOI: 10.1039/d3ta02323k.
- Witman, M.; Ek, G.; Ling, S. L.; Chames, J.; Agarwal, S.; Wong, J.; Allendorf, M. D.; Sahlberg, M.; Stavila, V. Data-Driven Discovery and Synthesis of High Entropy Alloy Hydrides with Targeted Thermodynamic Stability. Chemistry of Materials 2021, 33 (11), 4067-4076. DOI: 10.1021/acs.chemmater.1c00647.
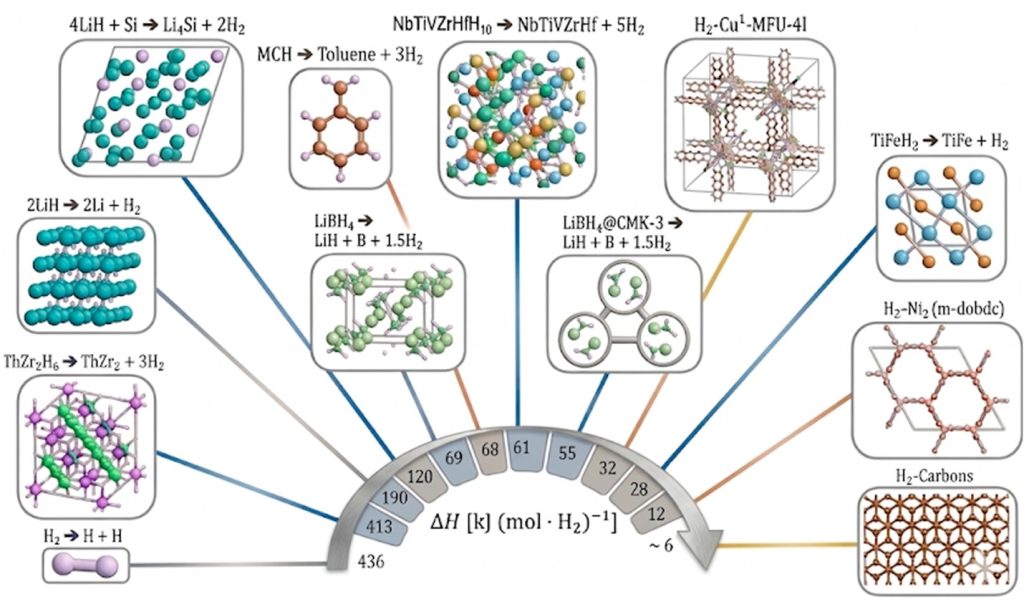
Hydrogen provides multiple advantages as an energy carrier, particularly when used in hydrogen polymer electrolyte membrane fuel cells. It is non-toxic, produces zero emissions at the point of use, and enables high efficiency conversion devices such as hydrogen fuel cells. Metal hydrides are promising solid-state hydrogen storage media, with record-high gravimetric and volumetric hydrogen densities. One of the most elegant and promising storage methods is through reversible metal hydrides, which can release hydrogen endothermically upon mild heating and absorb it exothermically when pressurized with hydrogen gas. Our research is geared toward developing a foundational understanding of the atomic to mesoscale phenomena that govern hydrogen uptake and release in sorbents, Liquid Organic Hydrogen Carriers, and metal hydrides, utilizing advanced diffraction and spectroscopic characterization tools to explore critical structure-property-function relationships. By employing a co-design strategy, we directly couple this fundamental materials discovery to systems modeling and techno-economic analysis, ensuring that our optimization of performance parameters like energy density, mass and thermal transport, and overall process efficiency meets the specific technical targets and requirements of hydrogen end-use cases.
Further Reading:
- Allendorf, M. D.; Stavila, V.; Snider, J. L.; Witman, M.; Bowden, M. E.; Brooks, K.; Tran, B. L.; Autrey, T. Challenges to developing materials for the transport and storage of hydrogen. Nature Chemistry 2022, 14 (11), 1214-+. DOI: 10.1038/s41557-022-01056-2.
- Charbonnier, V.; Witman, M. D.; Shinzato, K.; Kim, H.; Koh, J.; Cho, H.; Allendorf, M. D.; Asano, K.; Cho, E. S.; Sakaki, K.; Stavila, V. Performance assessment of AB2 alloys (A = Zr, Ti; B = Mn, Ni, Cr, V, Fe, Al) for hydrogen storage and separation in the presence of carbon dioxide. Journal of Alloys and Compounds 2026, 1055. DOI: 10.1016/j.jallcom.2026.186349.
- Taylor, W. V.; Klebanoff, L. E.; Ray, K. G.; Li, S. C.; Zhuo, Z. Q.; Kim, M. J.; Dun, C. C.; Johnson, A. C.; Marple, M. A. T.; El Gabaly, F.; Urban, J. J.; Guo, J. H.; Wood, B. C.; Stavila, V.; Allendorf, M. D. Decoding the Desorption Mechanism of 2LiH:1Mg(NH2)2 Using Metal Borohydrides. ACS Applied Energy Materials 2025, 8 (14), 10379-10391. DOI: 10.1021/acsaem.5c01297.
- Stavila, V.; Li, S. C.; Dun, C.; Marple, M. A. T.; Mason, H. E.; Snider, J. L.; Reynolds, J. E.; El Gabaly, F.; Sugar, J. D.; Spataru, C. D.; Zhou, X. W.; Dizdar, B.; Majzoub, E. H.; Chatterjee, R.; Yano, J.; Schlomberg, H.; Lotsch, B.; Urban, J. J.; Wood, B. C.; Allendorf, M. D. Defying Thermodynamics: Stabilization of Alane Within Covalent Triazine Frameworks for Reversible Hydrogen Storage. Angewandte Chemie-International Edition 2021, 60 (49), 25815-25824. DOI: 10.1002/anie.202107507.
- Snider, J. L.; Su, J.; Verma, P.; El Gabaly, F.; Sugar, J. D.; Chen, L. N.; Chames, J. M.; Talin, A. A.; Dun, C. C.; Urban, J. J.; Stavila, V.; Prendergast, D.; Somorjai, G. A.; Allendorf, M. D. Stabilized open metal sites in bimetallic metal-organic framework catalysts for hydrogen production from alcohols. Journal of Materials Chemistry A 2021, 9 (17), 10869-10881. DOI: 10.1039/d1ta00222h.
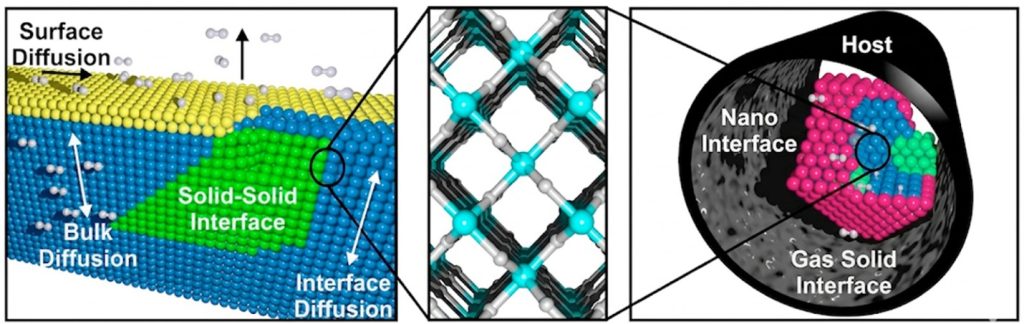
Nanoscale phenomena provide a route for altering material properties and achieving states unachievable by their bulk counterparts. We are interested in understanding how surfaces and nanointerfaces form and evolve, and how these changes translate into macroscopic property shifts. At nanoscale, interfaces can restructure and defects can accumulate, often altering mass and ion transport, and phase stability. These effects become especially pronounced under nanoconfinement within chemically functionalized pores, where materials are reduced to <10 nm in size. In this regime, local interfacial environments differ substantially from the bulk, governing thermodynamic stability, kinetic barriers, and reaction pathways in ways otherwise inaccessible. Purposeful control of nanointerfaces therefore provides a powerful route to tune physical and chemical properties. By tailoring pore size, surface functional groups, and interfacial composition, it becomes possible to influence adsorption, stabilize targeted intermediates, and promote desired pathways while suppressing competing reactions. A particularly promising example is the nanoconfinement of metal hydrides in porous hosts to tune thermodynamic stability and control the hydrogen to metal ratio, as recently demonstrated for metastable Al and La hydrides. This platform could enable the development of nanoscale superconducting materials, as well as hydrogen generation and storage materials. By exploring various species within functionalized porous architectures, we aim to establish quantitative design rules for how particle size, morphology, and local interfacial structure control properties across energy and fusion applications.
Further Reading:
- Shivanna, M.; Elmslie, T. A.; Spataru, C. D.; Duwal, S.; Porcellino, N.; Mouzaya, S.; Pakhanyan, C. P.; El Gabaly, F.; Kawaguchi-Imada, S.; Guo, J. H.; Zhuo, Z. Q.; Kim, M. J.; Sturtevant, B. T.; Teprovich, J. A.; Allendorf, M. D.; Sharma, P. A.; Stavila, V. Nanoconfinement of High Hydrogen-to-Metal Ratio Lanthanum Hydrides in Functionalized Carbon Hosts. ACS Applied Energy Materials 2024, 8 (1), 7-15. DOI: 10.1021/acsaem.4c02100.
- Dun, C.; Li, S. C.; Chen, L. F.; Horton, R. D.; Allendorf, M. D.; Wood, B. C.; Stavila, V.; Urban, J. J. A Nanoscale Ternary Amide-rGO Composite with Boosted Kinetics for Reversible H2 Storage. Advanced Materials Interfaces 2023, 10 (27). DOI: 10.1002/admi.202300310.
- Gunda, H.; Ray, K. G.; Klebanoff, L. E.; Dun, C.; Marple, M. A. T.; Li, S. C.; Sharma, P.; Friddle, R. W.; Sugar, J. D.; Snider, J. L.; Horton, R. D.; Davis, B. C.; Chames, J. M.; Liu, Y. S.; Guo, J. H.; Mason, H. E.; Urban, J. J.; Wood, B. C.; Allendorf, M. D.; Jasuja, K.; Stavila, V. Hydrogen Storage in Partially Exfoliated Magnesium Diboride Multilayers. Small 2023, 19 (6). DOI: 10.1002/smll.202205487.
- Stavila, V.; Li, S. C.; Dun, C.; Marple, M. A. T.; Mason, H. E.; Snider, J. L.; Reynolds, J. E.; El Gabaly, F.; Sugar, J. D.; Spataru, C. D.; Zhou, X. W.; Dizdar, B.; Majzoub, E. H.; Chatterjee, R.; Yano, J.; Schlomberg, H.; Lotsch, B.; Urban, J. J.; Wood, B. C.; Allendorf, M. D. Defying Thermodynamics: Stabilization of Alane Within Covalent Triazine Frameworks for Reversible Hydrogen Storage. Angewandte Chemie-International Edition 2021, 60, 25815-25824. DOI: 10.1002/anie.202107507.
- Schneemann, A.; White, J. L.; Kang, SY.; Jeong, S.; Wan, L. F.; Cho, E. S.; Heo, T. W.; Prendergast, D.; Urban, J. J.; Wood, B. C.; Allendorf, M. D.; Stavila, V. Chemical Reviews, 2018, 118, 10775-10839. ”Nanostructured Metal Hydrides for Hydrogen Storage”
Metal-Organic Frameworks (MOFs) are crystalline solids characterized by highly tunable structures, high surface areas, and large internal volumes. Although traditionally regarded as insulating, they can be rendered electronically or ionically conductive through the judicious choice of metal ions, linkers, and guest species confined inside the pores. Strict control over electronic and magnetic properties is crucial for integrating MOFs into quantum devices and sensors. Through various collaborations, we have contributed to the toolbox of techniques necessary to integrate MOF crystals and thin films into functional devices. Current research examines the spin behavior of transition metals in geometrically frustrated 2D and 3D lattices. By investigating how tunable hybrid materials with triangular, Kagome, or honeycomb lattices stabilize highly disordered spin states, hybridization and exchange coupling can be modulated. These systematic variations in metal ion composition lead to measurable changes in Curie Weiss behavior and heat capacity, providing a pathway to engineer quantum materials with precisely controlled magnetic properties.
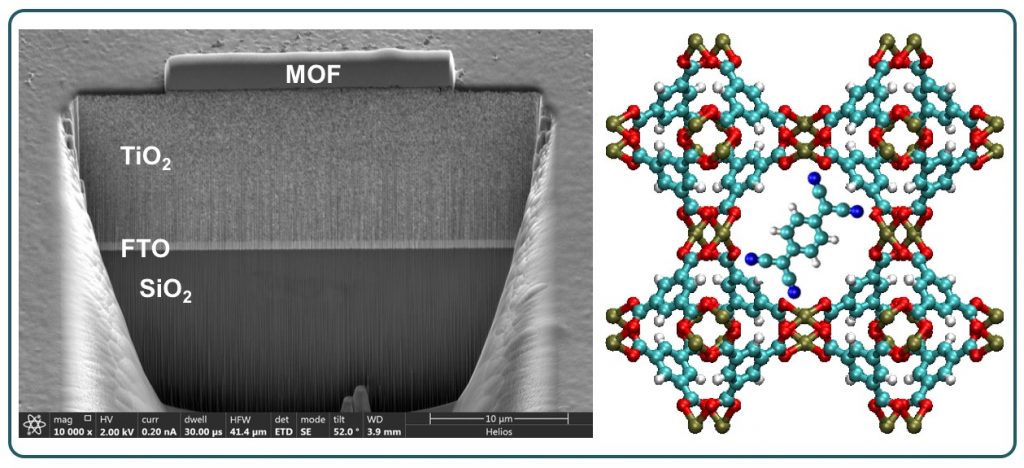
Further Reading:
- Shivanna, M.; Torquato, N. A.; Li, S. C.; Marple, M. A. T.; Sugar, J. D.; Foster, M. E.; Berry, A.; Taylor, W. V.; Strange, N. A.; Wang, X. L.; El Gabaly, F.; Chang, T. Y.; Chen, Y. S.; Sharma, P. A.; Lemmon, J.; Sadtler, B.; Wood, B. C.; Allendorf, M. D.; Stavila, V. Single-Electron Transfer Stabilizes Metastable Alane in a Bipyridine-Functionalized MOF Nanopore. Journal of the American Chemical Society 2025, 147, 47398-47408. DOI: 10.1021/jacs.5c15894.
- Yoon, S.; Talin, A. A.; Stavila, V.; Mroz, A. M.; Bennett, T. D.; He, Y. P.; Keen, D. A.; Hendon, C. H.; Allendorf, M. D.; So, M. C. From n- to p-Type Material: Effect of Metal Ion on Charge Transport in Metal-Organic Materials. Acs Applied Materials & Interfaces 2021, 13 (44), 52055-52062. DOI: 10.1021/acsami.1c09130.
- Timothy C. Wang, Patrick F. Doty, Annabelle Benin, Joshua D. Sugar, Vitalie Stavila, Mark D. Allendorf, Chemical Communications, 2019, 55, 4647-4650. “Get the light out: nanoscaling MOFs for luminescence sensing and optical applications.”
- V. Stavila, A.A. Talin, M.D. Allendorf, Chemical Society Reviews, 2014, 43, 5994-6010. “MOF-based electronic and opto-electronic devices.”
- A.A. Talin, A. Centrone, A.C. Ford, M.E. Foster, V. Stavila, P. Haney, R.A. Kinney, V. Szalai, F. El Gabaly, H.P. Yoon, F. Leonard, M.D. Allendorf, Science, 2014, 343, 66–69. “Tunable electrical conductivity in Metal-Organic Framework thin-film devices.”
Our approach to the design of highly active catalytic sites is based on principles of organometallic chemistry, biocatalysis, and computational chemistry. This enables rational design of novel homogeneous and heterogeneous catalysts with the ability to independently control the concentrations of Lewis and Brønsted acid sites, as well as hydricity of metal-hydrogen bonds for activation of various small molecules. The highly functionalized catalytic sites can stabilize reactive intermediates and prevent side reactions and deactivation. Through this methodology, we have successfully designed a range of multifunctional systems, including highly selective catalysts for C-O bond hydrogenolysis, CO2 hydrogenation, and reversible (de)hydrogenation of Liquid-Organic Hydrogen Carriers (LOHCs). Such catalysts could enable efficient processes for transformation of hydrogen and other small molecules into fuels and value-added chemicals.
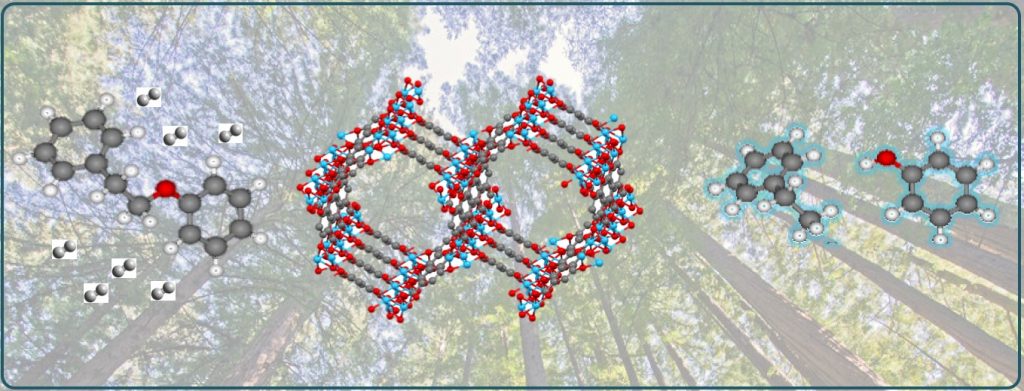
Further Reading:
- Hellman, A. N.; Torquato, N. A.; Foster, M. E.; Dun, C.; Reynolds, J. E., III; Yu, C. J.; Tran, A. D.; Shivanna, M.; Garcia, G. F. H.; Yang, J.; Chen, Y.; Su, J.; Urban, J. J.; Allendorf, M. D.; Stavila, V. Heterogenization of Homogeneous Ruthenium(II) Catalysts for Carbon-Neutral Dehydrogenation of Polyalcohols. ACS Applied Energy Materials 2023, 6 (14), 7353-7362. DOI: 10.1021/acsaem.3c00462.
- Stavila, V.; Foster, M.; Brown, J. W.; Davis, R. W.; Edgington, J.; Benin, A. I.; Zarkesh, R. A.; Parthasarathi, R.; Hoyt, D. W.; Walter, E. D.; Andersen, A.; Washton, N. M.; Lipton, A. S.; Allendorf, M. D., Chemical Science, 2019, DOI: 10.1039/C9SC01018A. “IRMOF-74(n)–Mg: a novel catalyst series for hydrogen activation and hydrogenolysis of C–O bonds.”
- V. Stavila, R. Parthasarathi, R.W. Davis, F. El Gabaly, K.L. Sale, B.A. Simmons, S. Singh, M.D. Allendorf, ACS Catalysis, 2016, 6, 55-59. “MOF-Based Catalysts for Selective Hydrogenolysis of Carbon–Oxygen Ether Bonds.”
The high cost of platinum group metals currently limits the broader adoption of electrochemical hydrogen conversion technologies. This challenge drives our research into high-entropy alloys and metallic glasses as earth abundant alternatives. These materials are specifically engineered for the Hydrogen Oxidation Reaction (HOR) and Hydrogen Evolution Reaction (HER), providing a robust foundation for future hydrogen fuel cell and electrolyzer designs. While replacing traditional precious metal electrocatalysts with less expensive metals typically reduces costs at the expense of efficiency, this project develops a fundamental understanding of the interactions between device components to maintain or even improve performance. By leveraging the unique, disordered atomic structures of these electrocatalysts, we can tailor a continuous distribution of active sites that match the catalytic activity of precious metals while offering exceptional corrosion resistance through self-limiting surface oxidation. Our approach utilizes an accelerated design loop that integrates artificial intelligence, atomistic modeling, and combinatorial synthesis. We employ machine learning derived interatomic potentials (MACE-MP) to predict key HOR and HER performance descriptors, such as hydrogen binding energy and work function, across thousands of potential alloy compositions. Validated through in situ and operando characterization, these efforts establish a roadmap for designing durable, earth-abundant electrocatalysts for the next generation of electrochemical energy conversion devices.
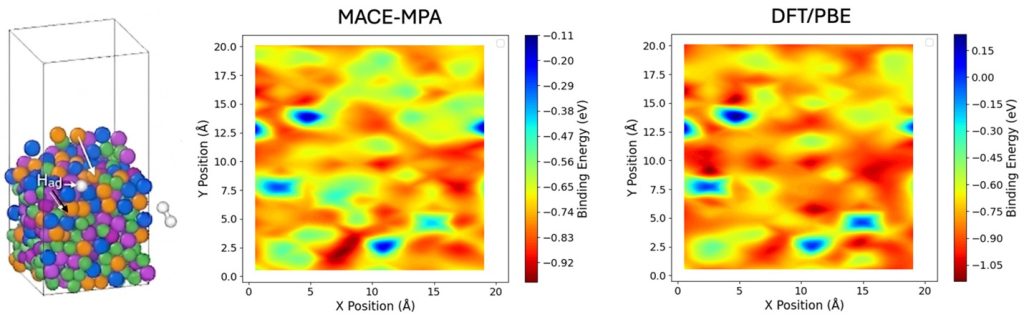
Further Reading:
- Bhagyashri Gaykwad, Donald A. Robinson, Nicholas Humphrey, Samuel F. Wenzel, Carly Hui, Natalia A. McCoy, Farid El Gabaly, Joshua D Sugar, Matthew David Witman, Kabeer Jasuja, Hang Ren and Vitalie Stavila, Composition-dependent hydrogen oxidation activity of Pt-Cu nanoparticles prepared using boron-rich nanosheets, Chem. Commun., 2026, DOI: 10.1039/D6CC00243A
- Tarancon, A.; Botros, M.; Gangwar, B. P.; Biswas, K.; Divilov, S.; Eckert, H.; Friedrich, R.; Calzolari, A.; Curtarolo, S.; Ludwig, A.; Strauss, F.; Brezesinski, T.; Breitung, B.; Fichtner, M.; He, R.; Cabot, A.; Dąbrowa, J.; Ren, J.-T.; Yuan, Z.-Y.; Zlotea, C.; et al. 2026 Roadmap on compositionally complex and high entropy materials for energy applications. Journal of Physics: Energy 2026, DOI:10.1088/2515-7655/ae57d9.
The majority of known solids are poor ionic conductors due to high activation energies and the lack of vacant sites for ion migration, which for a long time rendered their conductivity insufficient for battery applications. Very recently, however, the discovery of solids with ionic conductivities comparable to those of liquids has enabled the development of solid-state batteries with improved safety, voltage, and energy density. Within this context, we have developed a suite of solid-state electrolytes based on polyboron clusters that display superionic conductivity in a wide temperature range. Our studies show that cation disordering in these compounds originates from a competitive interplay among the lattice symmetry, the anion reorientational mobility, the geometric and electronic asymmetry of the anion, and the polarizability of the atoms decorating the polyboron cage. We use machine learning and data science to identify features responsible for high ionic conductivity and to suggest substitutions for optimizing cation-anion interactions. This provides valuable insights into the mechanisms responsible for fast ion transport, including the effects of confinement and structural disorder, and provides the practical design rules for the development of next generation superionic solid electrolytes.
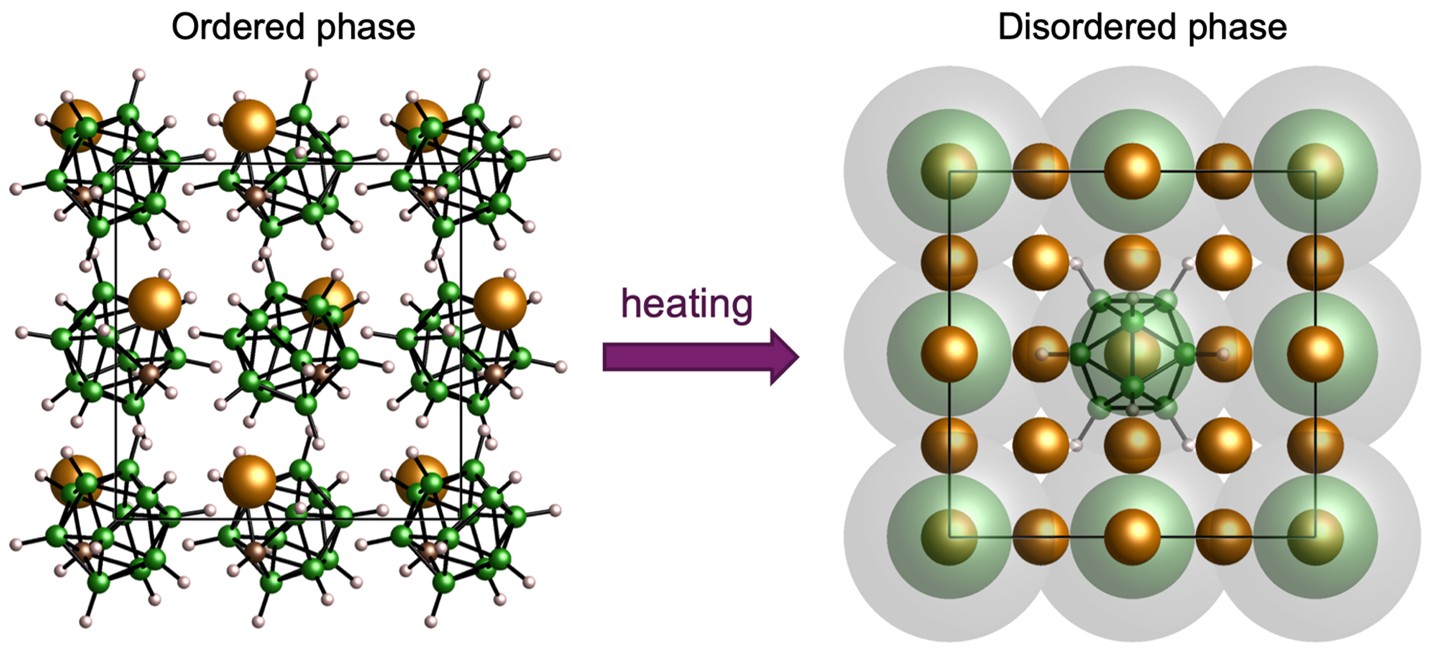
Further Reading:
- Jorgensen, M.; Shea, P. T.; Tomich, A. W.; Varley, J. B.; Bercx, M.; Lovera, S.; Cerny, R.; Zhou, W.; Udovic, T. J.; Lavallo, V.; Jensen, T. R.; Wood, B. C.; Stavila, V. Understanding Superionic Conductivity in Lithium and Sodium Salts of Weakly Coordinating Closo-Hexahalocarbaborate Anions. Chemistry of Materials 2020, 32, 1475-1487. DOI:10.1021/acs.chemmater.9b04383.
- Andersson, M. S.; Stavila, V.; Skripov, A.; Dimitrievska, M.; Psurek, M. T.; Leao, J. B.; Babanova, O. A.; Skoryunov, R.; Soloninin, A.; Karlsson, M.; Udovic, T. J. Promoting Persistent Superionic Conductivity in Sodium Monocarba-closo-dodecaborate NaCB11H12 via Confinement within Nanoporous Silica. Journal of Physical Chemistry C 2021, 125, 16689-16699. DOI: 10.1021/acs.jpcc.1c03589.
- Dimitrievska, M.; Shea, P.; Kweon, K. E.; Bercx, M.; Varley, J. B.; Tang, W. S.; Skripov, A. V.; Stavila, V.; Udovic, T. J.; Wood, B. C. Advanced Energy Materials 2018, 8, 1703422. ”Carbon Incorporation and Anion Dynamics as Synergistic Drivers for Ultrafast Diffusion in Superionic LiCB11H12 and NaCB11H12.”
- Tang, W. S.; Dimitrievska, M.; Stavila, V.; Zhou, W.; Wu, H.; Talin, A. A.; Udovic, T. J., Chemistry of Materials 2017, 29, 10496-10509. “Order-Disorder Transitions and Superionic Conductivity in the Sodium nido-Undeca(carba)borates.”
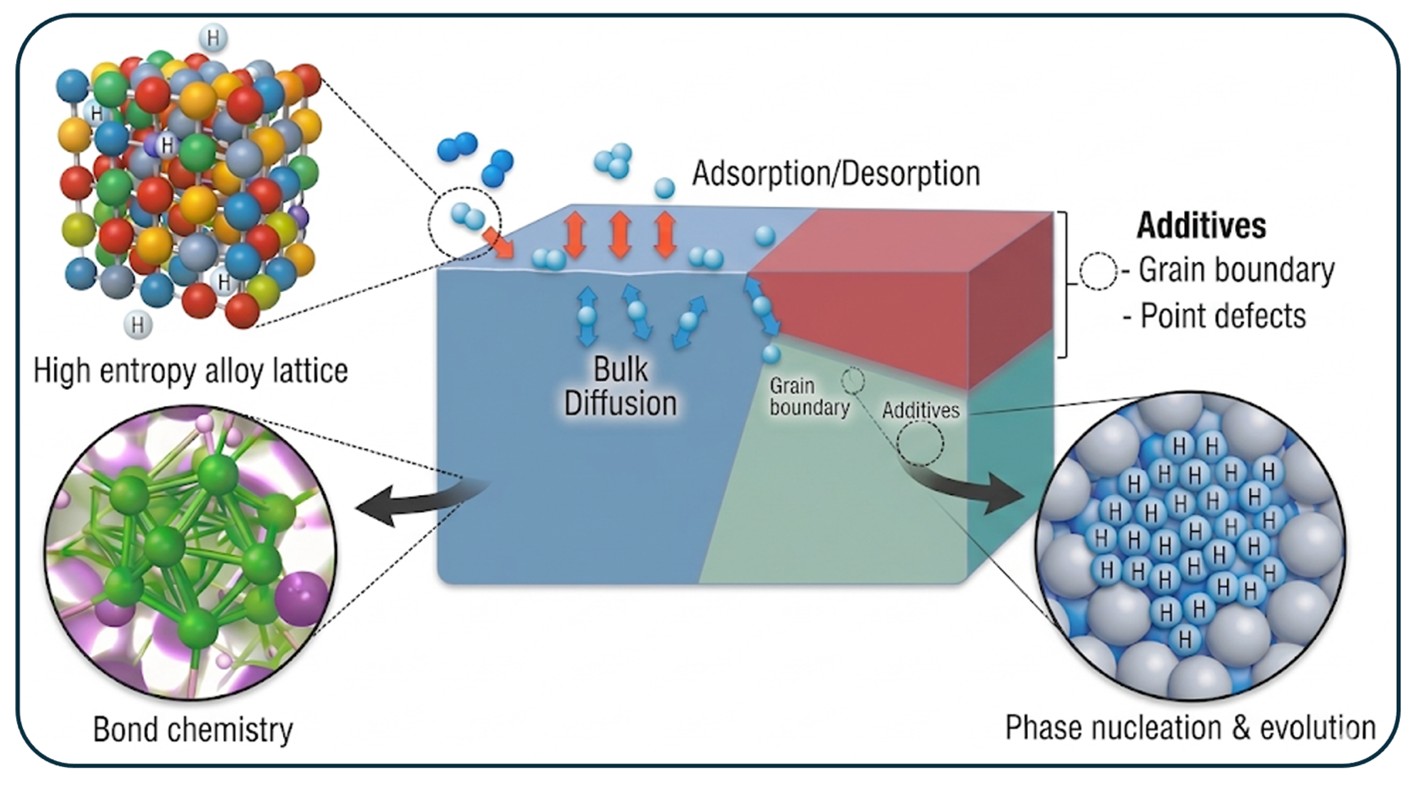
This research focuses on the design, synthesis, and fundamental characterization of advanced materials for next generation hydrogen separation technologies. At the heart of this effort is the exploration of novel materials such as high entropy alloys and metallic glasses, which offer a vast and largely untapped compositional space for tailoring hydrogen-material interactions. To navigate this complexity, we leverage AI guided robotic synthesis to precisely control composition and microstructure, allowing for the rapid and reproducible creation of materials with optimized properties. By elucidating the complex mechanisms governing hydrogen absorption, diffusion, and desorption, the group aims to develop membranes that significantly enhance the efficiency and reduce the cost of hydrogen infrastructure. A major thrust of the research involves understanding how microstructural characteristics and structural defects, including vacancies, dislocations, and grain boundaries, dictate a material’s performance and its susceptibility to hydrogen embrittlement. To address these challenges, our team integrates high throughput experimental techniques with data science and computational modeling, employing in-situ and operando methodologies to observe material behavior under operational conditions. This multidisciplinary approach enables the rational design of robust alloys that combine high hydrogen flux with the mechanical integrity required for long-term viability in gas purification applications.
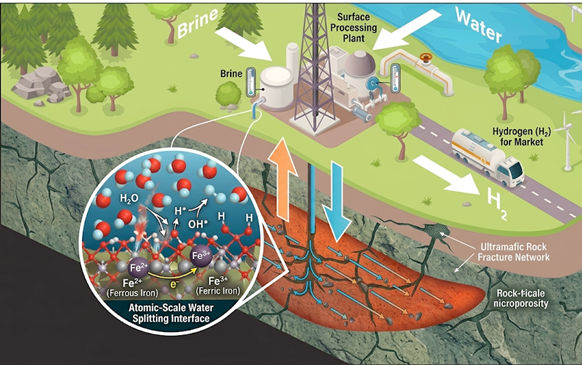
Leveraging Sandia’s specialized suite of high-pressure and high-temperature reactors, we are characterizing the kinetics of the serpentinization reaction and identifying the fundamental rate-limiting steps of hydrogen generation from Fe(II)-rich minerals such as fayalite, dunite, olivine, and peridotite. By subjecting these natural materials to pressures up to 100 MPa and temperatures between up to 500 °C, we effectively simulate the extreme geological conditions of the Earth’s crust to observe dihydrogen formation. This approach utilizes flow reactors, high-pressure DSC, and operando mass spectrometry to investigate how variables like particle size and trace catalytic elements can be strategically exploited to artificially accelerate the kinetics of the serpentinization process. Ultimately, these experiments bridge the gap between bench-top chemistry and deep-earth geophysics, providing the high-fidelity macroscopic data necessary to validate first-principles models and determine the feasibility of stimulating natural hydrogen production as a scalable energy source.
This effort leverages artificial intelligence and explainable machine learning tools to accelerate the discovery of next generation reversible hydrogen getters. By combining first-principles calculations with machine learning surrogate models, our team is developing interpretable design principles to predict the thermodynamics and kinetics of metal hydrides, deuterides, and tritides. The compositional model trained on an extensive experimental database evaluated a library of thousands of possible alloys and identified high entropy substitutions that tune plateau pressures and enhance impurity tolerance and cycle-life. A critical component of this work is the integration of legacy data regarding material aging and degradation, including the effects of surface segregation and lattice disorder on long-term durability and reaction kinetics. By bridging historical datasets with explainable AI, robotic synthesis, and advanced characterization, our goal is to establish a holistic understanding of synthesis-structure-property relationships and to develop a comprehensive database of materials optimized for various use cases in the hydrogen isotope lifecycle.