|
Distinguished Member of the Technical Staff
Bioenergy and Defense Technologies Department
(505) 844-7932
jatimli@sandia.gov
|
Research Interests
Optical microscopy, spectroscopy, and spectral imaging (including fluorescence, Raman, and Fourier transform infrared [FTIR]), high-throughput imaging, superresolution techniques, multiplexed microscopy, multivariate image analysis, image preprocessing, spectral unmixing, and multivariate classification for applications in the early detection of disease/damage, immune response, signal transduction, energy transfer in photosynthetic pigments, and biomarker discovery.
|
|
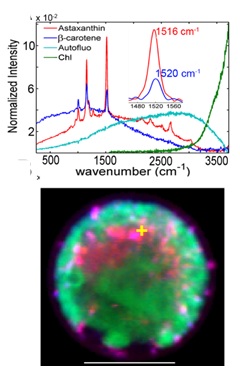
My research focuses on developing and applying novel analytical-imaging and multivariate-analysis tools to elucidate complex spatial-temporal relationships of a variety of biomolecules that drive key biological processes. The work in our lab crosses traditional boundaries of chemistry, physics, and biology and often covers multiple spatial scales from single molecules on up to single cells, communities, and tissues. A main component of our work is spectral imaging - a method whereby spectrally resolved information is obtained at every two-dimensional pixel or three dimensional voxel. I have been developing and applying various spectral imaging technologies (also called chemical imaging) since my graduate work which pioneered the use of hyperspectral Raman microscopy with near-infrared excitation to elucidate the dynamic chemical composition of bone without the use of labels. Most recently, my group has employed both hyperspectral Raman and fluorescence microscopy to a variety of applications looking at single molecules to intact tissue, including the visualization of subcellular pigment distribution in photosynthetic organisms such as cyanobacteria and green algae (Figure 1).
The addition of a spectral dimension can result in a three-, four-, or five-dimension image data set (2-3 spatial, 1 spectral, and 1 temporal) that is beyond human visualization capabilities. For this reason we utilize sophisticated multivariate analysis tools to mathematically extract the underlying spectral signatures and create quantitative spatial-temporal profiles of biomolecules.
In keeping with the multicolor theme, our lab has developed multi-color, optical super resolution capabilities. Building from our unique capabilities in dual-color, video rate, total internal reflection fluorescence (TIRF) microscopy, we have constructed a simultaneous, dual-color single molecule, stochastic optical super-resolution (STORM) microscope. With spatial resolutions approaching those of electron microscopy (~30-40 nm), this technology is opening our eyes to a variety of biological processes never before seen. For example, Figure 2 highlights receptor reorganization at the membrane during immune response. |
Figure 1: Hyperspectral confocal Raman image of carotenoids and chlorophyll in living Haematococcus pluvialis cells. Upper panel: component spectra. Lower panel: pigment localization, pseudo-colored corresponding to spectra in upper panel. Figure generated by Aaron Collins in collaboration with Thomas Beechem, and Howland Jones at SNL and Dr. Qiang Hu's group at ASU.
|
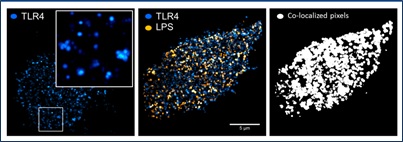 |
Figure 2: Nanoscale organization of Toll-like Receptor 4 (TLR4) and E. coli lipopolysaccharide (LPS) at the plasma membrane of mouse macrophage cells as visualized with simultaneous, dual-color stochastic optical reconstruction microscopy (STORM). Figure generated by Jesse Aaron, work in collaboration wtih Bryan Carson at SNL. |
Our work interfaces analytical chemistry and optical physics with molecular and cellular biology and has direct impact on human health, plant science, and renewable energy research. We work collaboratively with researchers from different scientific disciplines to tackle complex bioscience problems.
Top of page ^
Multiplexed Measurements of Protein Dynamics and Interactions at Extreme Resolutions
This project was funded under the National Institutes of Health (NIH) Director’s New Innovator Award in 2009. When successful, this project will provide an unprecedented view of protein interactions in the living cell through the development of novel spectrally resolved superresolution microscopy methods.
Analyzing and Understanding of Transporters to Control Lignin Transformation into Fuel
The overall goal of this project is to understand the fundamental relationships that govern lignin transport in order to control transport in a way that will provide an increase in the range and kinetics of transport into cells. We will develop analytical tools for assessing lignin uptake and kinetics in bulk and at the single cell/single molecule level. Combining experimental measurements with computational modeling, we hope to identify the types of transporters are active on lignin and their substrate specificity as well the kinetics of lignin transport in natural and engineered hosts.
PARC is one of 46 Energy Frontier Research Centers (EFRCs) established nationally at universities, national laboratories, nonprofit organizations, and private firms by the U.S. Department of Energy’s Office of Science. A multi-institutional collaboration, PARC seeks to understand the basic scientific principles that underpin the efficient functioning of natural photosynthetic antenna systems. These principles will then be used as a basis for man-made systems to convert sunlight into fuels. Our specific work within PARC will use advanced spectral imaging and analysis methodologies to isolate fluorescent signatures from natural and bio-inspired photosynthetic pigments to increase our understanding of the spatial distribution and abundance of these critical components in the energy transfer cascade.
Top of page ^
Timlin, JA, Collins, AM, Shumskaya, M, Wurtzel, ET, Beechem, TA, Localizing and Quantifying Carotenoids in Intact Cells and Tissues in Carotenoids. Cvetkovic, D and Nikolic, G. In Tech, 2017, in press.
Anthony S, Carroll-Portillo A and Timlin JA, "Dynamics and interactions of individual proteins in the membrane of single, living cells " in Single Cell Protein Analysis. Singh, AK and Chandrasekaran, A, editor(s). Springer New York: 2015, 185-207.
Chen W, Han D, Li Y, Jones HDT, Timlin JA, Hu Q, Semi-Quantitative and Absolute Quantitative Analyses of Biochemical Composition of Microalgae. In Handbook of Microalgal Cultures - Second Edition. Hu R, (Ed.), Wiley-Blackwell, 2013.
Aaron JS and Timlin JA Advanced Optical Imaging of Endocytosis, in Molecular Recognition of Endocytosis, B. Ceresa (Ed.), InTech, 2012.
Timlin JA, "Scanning microarrays: Current methods and future directions," In DNA Microarrays, Part B: Databases and Statistics. Kimmel A, Oliver B, editors. Academic Press: New York, 2006: 79-98.
Top of page ^
2014 – present
- MacGregor-Chatwina, C, Sener, M, Barnett, SFH, Hitchcock, A, Barnhart-Dailey, MC, Maghlaoui, K, Barber, J, Timlin, JA, Schulten, K and Hunter, CN, "Lateral segregation of photosystem i in cyanobacterial thylakoids," The Plant Cell, 2017, in press.
- Collins, AM, Timlin, JA, Anthony, SM and Montano, G, "Amphiphilic block copolymers as flexible membrane materials generating structural and functional mimics of green bacterial antenna complexes," Nanoscale, 2016, 8:15056.
- Anthony, SM and Timlin, JA, "Removing cosmic spikes using a hyperspectral upper-bound spectrum method," Applied Spectroscopy, 2016, 71:3, 507-519.
- Toomey, MB, Collins, AM, Frederiksen, R, Cornwall, MC, Timlin, JA and Corbo, JC, "A complex carotenoid palette tunes avian color vision," Journal of the Royal Society Interface, 2015, 12:111.
- Kent, M, Avina, IC, Rader, N, Busse, M, George, A, Sathitsuksanoh, N, Baidoo, E, Timlin, JA, Giron, N, Celina, M, Chavez, V, Polsky, R, Huber, D, Martin, LE, Keasling, J, Singh, S, Simmons, BA and Sale, KL, "New assay for lignin breakdown based on lignin films: Insights into the fenton reaction with insoluble lignin," Green Chemistry, 2015, 17, 4830-4845.
- Anthony, S, Carroll-Portillo, A and Timlin, J, "Dynamics and interactions of individual proteins in the membrane of single, living cells quot; in Single Cell Protein Analysis. Singh, AK and Chandrasekaran, A, editor(s). Springer New York: 2015, 185-207.
- Hanson DT, Collins AM, Jones HDT, Roesgen J, Lopez-Nieves S, Timlin JA, "On-line stable isotope gas exchange reveals an inducible but leaky carbon concentrating mechanism in Nannochloropsis salina," Photosynthesis Research, 2014, 121:311-322.
- Collins AM, Jones HDT, McBride RC, Behnke, CA, Timlin JA, "Host cell pigmentation in Scenedesmus dimorphus as a beacon for nascent parasite infection," Biotechnology and Bioengineering, 2014, 111:9, 1748-1757.
- Reichardt TA, Collins AM, McBride RC, Behnke, CA, Timlin, JA, "Spectroradiometric monitoring for open outdoor culturing of algae and cyanobacteria," Applied Optics, 2014, 53:24, F31-F45.
2011 – 2013
- Chen W, Han D, Li Y, Jones HDT, Timlin JA, Hu Q, Semi-Quantitative and Absolute Quantitative Analyses of Biochemical Composition of Microalgae. In Handbook of Microalgal Cultures - Second Edition. Hu R, (Ed.), Wiley-Blackwell, 2013 - in press.
- Davis RW, Jones HDT, Collins AM, Ricken JB, Sinclair MB, Timlin JA and Singh S. "Label-free measurement of algal triacylglyceride production using fluorescence hyperspectral imaging," Algal Research, 2013, 5, 181-189.
- Kilcrease, JA, Collins, AM, Richins, RD, Timlin, JA and O'Connell, MA, "Multiple microscopic approaches demonstrate linkage between chromoplast architecture and carotenoid composition in diverse capsicum annum fruit," The Plant Journal, 2013, 76:6, 1074-1083.
- Liberton M, Collins A, Page L, O'Dell W, O'Neill H, Urban V, Timlin JA, Pakrasi, HB, "Probing the consequences of antenna modification in cyanobacteria," Photosynthesis Research, 2013, 1-8.
- Aaron, JS and Timlin JA "Advanced Optical Imaging of Endocytosis," in Molecular Recognition of Endocytosis, B. Ceresa (Ed.), InTech, 2012.
- Aaron JS, Carson BD, Timlin JA "Characterization of Differential Toll-Like Receptor Responses below the Optical Diffraction Limit," Small, 2012, 8(19), 3041-3049.
- Jones HDT, Haaland DM, Sinclair MB, Melgaard DK, Collins A M, Timlin JA "Preprocessing Strategies to Improve MCR Analyses of Hyperspectral Images," Journal of Chemometrics and Intelligent Laboratory Systems, 2012, 117, 149-158.
- Collins, AM, Liberton, M, Jones, HDT, Garcia, OF, Pakrasi, HB, Timlin, JA "Photosynthetic Pigment Localization and Thylakoid Membrane Morphology are Altered in Synechocystis 6803 Phycobilisome Mutants" Plant Physiology, 2012, 158, 1600-1609.
- Reichardt TA, Collins AM, Garcia OF, Ruffing AM, Jones HDT, Timlin JA "Spectroradiometric Monitoring of Nannochloropsis salina Growth," Algal Research, 1(1), 2012, 22-31.
- Collins AM, Jones HDT, Han D, Hu Q, Beechem TE, and Timlin JA "Carotenoid Distribution in Living Cells of Haematococcus pluvialis (Chlorophyceae)," PLoS One, 6(9), 2011, e24302.
- Aaron JS, Greene A Kotula PG, Bachand GD, and Timlin JA. "Advanced Optical Imaging Reveals Dependence of Particle Geometry on Interactions between CdSe Quantum Dots and Immune Cells." Small, 7(3), 2011, 334-341.
Top of page ^
2008 –2010
- Davis, RW, JA Timlin, R Noek, JN Kaiser, HDT Jones, TW Lane. (2010). Accurate detection of low levels of fluorescence emission in autofluorescent background: Francisella-infected macrophage cells. Microscopy & Microanalysis 16:478.
- Spendier, K, A Carroll-Portillo, KA Lidke, BS Wilson, JA Timlin, JL Thomas. (2010). Distribution and dynamics of rat basophilic leukemia immunoglobulin E receptors (FcεRI) on planar ligand-presenting surfaces. Biophysical Journal 99:388.
- Carroll-Portillo, A, K Spendier, K Lidke, J Pfeiffer, D Lidke, J Thomas, B Wilson, JA Timlin. (2010). Formation of a mast cell synapse: FcεRI membrane dynamics upon binding mobile or immobilized ligands on surfaces. Journal of Immunology 184:1328.
- Thomas, EV, KH Phillippy, B Brahamsha, DM Haaland, JA Timlin, B Palenik, I Paulsen. (2009). Statistical analysis of microarray data with replicated spots: A case study with Synechococcus WH8102. Comparative and Functional Genomics Article ID 950171.
- Timlin, JA, LE Martin, CR Lyons, B Hjelle, MK Alam. (2009). Dynamics of cellular activation as revealed by attenuated total reflectance infrared spectroscopy. Vibrational Spectroscopy 50:78.
- Vermaas, WJ, JA Timlin, HDT Jones, MB Sinclair, LT Nieman, S Hamad, DK Melgaard, DM Haaland. (2008). In vivo hyperspectral confocal fluorescence imaging to determine pigment localization and distribution in cyanobacterial cells. Proceedings of the National Academy of Sciences 105:4050.
Top of page ^
2004 – 2007
- Alam, MK, JA Timlin, LE Martin, D Williams, CR Lyons, B Hjelle. (2004). Classification of mouse macrophage cells using attenuated total reflectance infrared spectroscopy. Vibrational Spectroscopy 34:3.
- Guzowski, JF, JA Timlin, B Roysam, BL McNaughton, PF Worley, CA Barnes. (2005). Mapping behaviorally relevant neural circuits with immediate-early gene expression. Current Opinion in Neurobiology 15:599.
- Sinclair, MB, DM Haaland, JA Timlin, HDT Jones. (2006). Hyperspectral confocal microscope. Applied Optics 45:3283.
- Sinclair, MB, JA Timlin, DM Haaland, M Werner-Washburne. (2004). Design, construction, characterization, and application of a hyperspectral microarray scanner. Applied Optics 43:2079.
- Sutherland, V, JA Timlin, LT Nieman, JF Guzowski, MK Chawla, B Roysam, PF Worley, BL McNaughton, MB Sinclair, CA Barnes. (2007). Advanced imaging of multiple mRNAs in brain tissue using a custom hyperspectral imager and multivariate curve resolution. Journal of Neuroscience Methods 160:144.
- Timlin, JA. (2006). Scanning microarrays: Current methods and future directions. In DNA Microarrays, Part B: Databases and Statistics, A Kimmel, B Oliver, Eds. Academic Press: New York, pp. 79–98.
- Timlin, JA, DM Haaland, MB Sinclair, AD Aragon, MJ Martinez, M Werner-Washburne. (2005). Hyperspectral microarray scanning: Impact on the accuracy and reliability of gene expression data. BMC Genomics 6:72.
Top of page ^
1999 – 2003
- Heffelfinger, GS, A Martino, AY Gorin, Y Xu, MD Rintoul III, A Geist, HM Al-Hashimi, GS Davidson, JL Faulon, LJ Frink, DM Haaland, WE Hart, E Jakobsson, T Lane, M Li, P Locascio, F Olken, V Olman, B Palenik, SJ Plimpton, DC Roe, NF Samatova, M Shah, A Shoshoni, CEM Strauss, EV Thomas, JA Timlin, D Xu. (2002). Carbon sequestration in Synechococcus sp.: From molecular machines to hierarchical modeling. OMICS: A Journal of Integrative Biology 6:305.
- Martinez, MJ, AD Aragon, AL Rodriguez, JM Weber, JA Timlin, MB Sinclair, DM Haaland, M Werner-Washburne. (2003). Identification and removal of contaminating fluorescence from commercial and in-house printed DNA microarrays. Nucleic Acid Research 31:e18.
- Timlin, JA, A Carden, MD Morris. (1999). Chemical microstructure of cortical bone probed by Raman transects. Applied Spectroscopy 53:1429.
- Timlin, JA, A Carden, MD Morris, JF Bonadio, CE Hoffler II, KM Kozloff, SA Goldstein. (1999). Spatial distribution of phosphate species in mature and newly generated mammalian bone by hyperspectral Raman imaging. Journal of Biomedical Optics 4:28.
- Timlin, JA, A Carden, MD Morris, RM Rajachar, DH Kohn. (2000). Raman spectroscopic imaging markers for fatigue-related microdamage in bovine bone. Analytical Chemistry 72:2229.
| 2002 |
PostDoc, Analytical Chemistry, Sandia National Labs, Albuquerque, NM |
| 2000 |
PhD, Analytical Chemistry, University of Michigan, Ann Arbor, MI |
| 1995 |
BS, Chemical Engineering, Geneva College, Beaver Falls, PA |
Top of page ^

Meghan Dailey
Postdoctoral Apppointee
Meghan received her B.S. in Biochemistry from the University of New Mexico in 2008. She attended the University of Virginia for her doctoral studies where she received her Master's and Ph.D. in Biochemistry and Molecular Genetics in 2010 and 2014, respectively. She did her thesis research in Dan Foltz's lab (now at Northwestern University) studying centromeric chromatin establishment and maintenance. She was the recipient of an NIH Molecular Biology Training Grant and UVA Farrow Fellowship, supporting her salary for the first 4 years of her doctoral studies. Upon completion of her doctoral work, she continued in the Foltz lab for 2 years as a post-doctoral fellow studying the role of the Condensin II complex on centromeric chromatin establishment. She then moved to Sandia National Laboratories in 2016 to finish her post-doctoral training under Jeri Timlin in the Biofuel and Biodefense Systems department. She is currently studying lignin transport in biofuel-relevant organisms using LC/MS, super-resolution, confocal, and TIRF microscopy as her primary project. She also performs hyperspectral imaging studies of mammalian, algal, and cyanobacterial cells, with applications ranging from photosynthetic pigment studies to TB drug compound testing. She and her husband enjoy time hiking and running in the sunny New Mexico weather and are looking forward to the arrival of their first baby in April 2017.
Publications
Lateral segregation of photosystem I in cyanobacterial thylakoids.
Craig MacGregor-Chatwin, Melih Sener, Samuel Barnett, Andrew Hitchcock, Meghan Barnhart-Dailey, Karim Maghlaoui, James Barber, Jerilyn Timlin, Klaus Schulten, and Christopher Hunter.
The Plant Cell. 2017, In press.
HJURP interaction with the condensin II complex during G1 promotes CENP-A deposition.
Barnhart-Dailey MC, Trivedi P, Stukenberg PT, Foltz DR.
Mol Biol Cell. 2017 Jan 1;28(1):54-64. doi: 10.1091/mbc.E15-12-0843.
Centromere licensing: Mis18 is required to Polo-ver.
Barnhart-Dailey MC, Foltz DR.
Curr Biol. 2014 Sep 8;24(17):R808-10. doi: 10.1016/j.cub.2014.07.026.
Dimerization of the CENP-A assembly factor HJURP is required for centromeric nucleosome deposition.
Zasadzinska E, Barnhart-Dailey MC, Kuich PH, Foltz DR.
EMBO J. 2013 Jul 31;32(15):2113-24. doi: 10.1038/emboj.2013.142.
HJURP uses distinct CENP-A surfaces to recognize and to stabilize CENP-A/histone H4 for centromere assembly.
Bassett EA, DeNizio J, Barnhart-Dailey MC, Panchenko T, Sekulic N, Rogers DJ, Foltz DR, Black BE.
Dev Cell. 2012 Apr 17;22(4):749-62. doi: 10.1016/j.devcel.2012.02.001.
HJURP is a CENP-A chromatin assembly factor sufficient to form a functional de novo kinetochore.
Barnhart MC1, Kuich PH, Stellfox ME, Ward JA, Bassett EA, Black BE, Foltz DR .
J Cell Biol. 2011 Jul 25;194(2):229-43. doi: 10.1083/jcb.201012017. Epub 2011 Jul 18.
Top of page ^