 |
Post Doctoral Researcher
|
|
Applied spectroscopy Advanced optical microscopies Biofuels |
Aaron Collins is a 2010 Ph.D. graduate from the Washington University Department of Chemistry. Aaron's Ph.D. work focused on using optical spectroscopies and analytical biochemistry to determine the structural and functional properties of photosynthetic complexes from so-called green bacteria. He joined the Timlin group in 2010 to investigate the distribution of natural photosynthetic complexes in living cells and how the global architecture relates to function as part of the Photosynthetic Antenna Research Center (PARC). His other research interests include Raman and fluorescence microscopy investigations of health and productivity in biofuels-relevant algae.
We have investigated how cyanobacteria remodel their thylakoid membrane architecture in response to a diminished light-harvesting antenna and have shed-light on the global distribution of PSII and PSI within living cells.
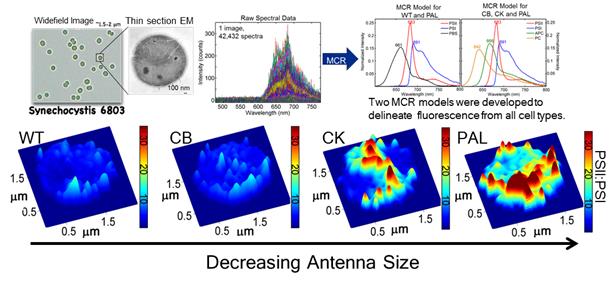
Hyperspectral confocal fluorescence microscopy was used in conjunction with multivariate curve resolution to separate PSI, PSII and phycobilisome emission from highly-congested fluorescence spectral images (top panel). Note as the phycobilisome antenna was diminished, increased segregation of the two photosystems become apparent at the single cell level (bottom panel).
Confocal Raman microscopy and multivariate curve resolution (MCR) can be used to provide molecular information on the microscopy scale, particularly for non-fluorescent biomolecules. Here, we used these techniques to deconfolute spectral signatures for astaxanthin, β-carotene to astaxanthin occurs in lipid vesicles outside of the chloroplast. |
|
Unicellular green algae harness solar energy using carotenoids, Chl a, and Chl b. We investigated wild-type and mutant lines of Chlamydomonas reinhardtii with truncated light-harvesting antennas to probe how decreasing Chl content influences global architecture of the chloroplast and pigment distribution. MCR analysis of these cell lines yields two spectral components representing LHCII and PSII. |
|
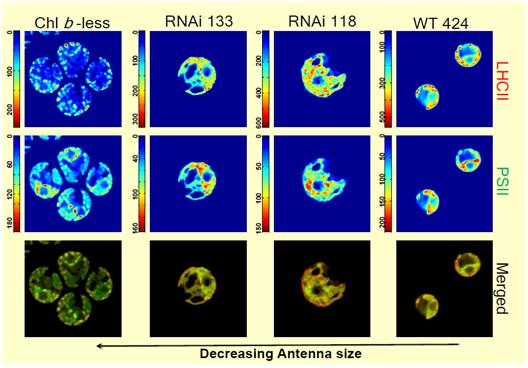
Wild-type cells show typical chloroplast morphology. LHCII and PSII were generally co-located; however, the chloroplast periphery appeared to be enriched in LHCII and the interconnecting regions had more PSII. The Chl b-less mutant was largely devoid of LHCII and the chloroplast morphology had regions that were punctate. The two intermediate-sized antenna mutants (RNAi118 and RNAi133) had spectral component distributions similar to wild-type cells; however, the overall chloroplast morphology was disorganized. In all cell lines, the concentration of PSII is equivalent. |
|
Collaboration with Sayre Group of the New Mexico Consortium
Jones HDT, Haaland DM, Sinclair MB, Melgaard DK, Collins AM, and Timlin JA (2012). Preprocessing Strategies to Improve MCR Analyses of Hyperspectral Images. Chemom. Intell. Lab. Syst. doi:10.1016/j.chemolab.2012.01.011.
Reichardt TA, Collins AM, Garcia OF, Ruffing AM, Jones HDT, and Timlin JA (2012). Spectroradiometric monitoring of Nannochloropsis salina growth. Algal Res. doi:10.1016/j.algal.2011.12.001.
Collins AM, Liberton M, Garcia OF, Jones HDT, Pakrasi HB, and Timlin JA (2012). Photosynthetic pigment localization and thylakoid membrane morphology are altered in Synechocystis 6803 phycobilisome mutants. Plant Phys. doi:10.1104/pp.111.192849.
Collins AM, Jones HDT, Han D, Hu Q, Beechem TE, and Timlin JA (2011). Carotenoid distribution in living cells of Haematococcus pluvialis (Chlorophyceae). PLoS ONE 6: e24302.
Xin Y, Pan J, Collins AM, Lin S, and Blankenship RE (2011). Excitation energy transfer and trapping dynamics in the core complex of the filamentous photosynthetic bacterium Roseiflexus castenholzii. Photosyn. Res. doi: 10.1007/s11120-011-9669-6.
Brotosudarmo THP, Collins AM, Gall A, Roszak AW, Gardiner AT, Blankenship RE, and Cogdell RJ (2011). The light intensity at which cells are grown controls the type of peripheral light-harvesting complexes that are assembled in a purple photosynthetic bacterium. Biochem. J. doi: 10.1042/BJ20110575.
Tang KH, Zhu Y, Urban VS, Collins AM, Biswas P and Blankenship RE (2011). Temperature and ionic strength effects on the chlorosome light-harvesting antenna complex. Lagmuir. 27, 4816-4828.
Collins AM, Kirmaier C, Holten D, and Blankenship RE (2011). Kinetics and Energetics of Electron Transfer in Reaction Centers of the Photosynthetic Bacterium Roseiflexus castenholzii. Biochim. Biophys. Acta. 1807, 262-269.
Collins AM, Qian P, Tang Q, Bocian DF, Hunter CN, and Blankenship RE (2010). Light-Harvesting Antenna System from the Phototrophic Bacterium Roseiflexus castenholzii. Biochemistry. 49, 7524-7531.
Niedzwiedzki DM, Collins AM, Lafountain AM, Enriquez MM, Frank HA, and Blankenship RE (2010), Spectroscopic Studies of Carotenoid-to-Bacteriochlorophyll Energy Transfer in LHRC Photosynthetic Complex from Roseiflexus castenholzii. J Phys Chem B. 114, 8723-8734.
Collins AM, Kevin E, Redding KE and Blankenship RE (2010). Fluorescence Modulation in Heliobacterium modesticaldum. Photosyn. Res. 104, 283-292.
Modesto-Lopez LB, Thimsen EG, Collins AM, Blankenship RE, and Biswas P (2010). Electrospray-assisted Characterization and Deposition of Chlorosomes to Fabricate a Light-Harvesting Nano-bio Device. Energy Environ. Sci., 3, 216-222.
Moulisova V, Luer L, Hoseinkhani S, Brotosudarmo THP, Collins AM, Lazani G, Blankenship RE, and Cogdell RJ (2009), Low light adaptation: Energy Transfer Processes in Different Types of Light Harvesting Complexes from Rhodopseudomonas palustris. Biophys. J. 97, 3019-3028.
Psencik J, Collins AM, Liljeroos L, Torkkeli M, Laurinmaki P, Ansink HM, Ikonen TP, Serimaa RE, Blankenship RE, Tuma R, and Butcher SJ (2009). Structure of the Chlorosomes from Green Filamentous Bacterium Chloroflexus aurantiacus. J. Bacteriology, 191, 6701-6708.
Collins AM, Xin Y, Blankenship RE (2009). Pigment Organization in the Photosynthetic Apparatus of Roseiflexus castenholzii. Biochim. Biophys. Acta. 1787, 1050-1056.
van de Meene AML, Olson TL, Collins AM and Blankenship RE (2007). Initial Characterization of the Photosynthetic Apparatus of "Candidatus Chlorothrix halophila": A Filamentous, Anoxygenic Photoautotroph. J. Bacteriology 189: 4196-4203.
Collins AM, Wen J, and Blankenship RE (2012). Photosynthetic Light-Harvesting Complexes. In TJ Wydrzynshi, W Hillier, eds, Molecular Solar Fuels. The Royal Society of Chemistry, Cambridge, pp 85-106.