World’s smallest battery created at CINT nanotechnology center
By Neal Singer
A benchtop version of the world’s smallest battery — its anode a single nanowire one seven-thousandth the thickness of a human hair —has been created by a team led by Jianyu Huang (1132).
To better study the anode’s characteristics, the tiny rechargeable, lithium-based battery was formed inside a transmission electron microscope (TEM) at the Center for Integrated Nanotechnologies (CINT), a DOE research facility jointly operated by Sandia and Los Alamos national laboratories.
THREADING THE NEEDLE Jianyu Huang demonstrates insertion of a sample holder into a transmission electron microscope at CINT. (Photo by Randy Montoya)
“These are the first controlled measurements of the growth of magneto-Rayleigh-Taylor [MRT] instabilities in fast Z-pinches,” says project lead Daniel Sinars (1643).
Says Jianyu of the work, reported in the Dec. 10 Science, “This experiment enabled us to study the charging and discharging of a nanobattery in real time and at atomic-scale resolution, thus enlarging our understanding of the fundamental mechanisms by which batteries work.”
Because nanowire-based materials in lithium ion batteries offer the potential for significant improvements over bulk electrodes in power and energy density, more stringent investigations of their operating properties should improve new generations of plug-in hybrid electric vehicles, laptops, and cell phones.
“What motivated our work,” says Jianyu, “is that lithium ion batteries [LIB] have very important applications, but the low energy and power densities of current LIBs cannot meet the demand. To improve performance, we wanted to understand LIBs from the bottom up, and we thought in-situ TEM could bring new insights to the problem.”
Battery research groups around the world use nanomaterials as anodes, but in bulk rather than individually — a process, Jianyu says, that resembles “looking at a forest to try to understand the behavior of an individual tree.”
The Medusa twist: formerly unobserved increase in length and twist of the anode in a nanobattery. Click the image to view a video showing the in-situ visualization of the battery charging process, a view never before possible.( image courtesy DOE Center for Integrated Nanotechnologies)
Observing change in atomic structure
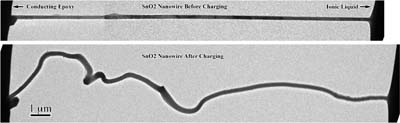
The Sandia-led design consists of a single tin oxide nanowire anode 100 nanometers in diameter and 10 micrometers long, a bulk lithium cobalt oxide cathode three millimeters long, and an ionic liquid electrolyte. The device offers the ability to directly observe change in atomic structure during charging and discharging of the individual “trees.”
An unexpected find of the researchers was that the tin oxide nanowire rod nearly doubles in length during charging — far more than its diameter increases — a fact that could help avoid short circuits that shorten battery life. “Manufacturers should take account of this elongation in their battery design,” Jianyu says. (The common belief of workers in the field had been that batteries swell across their diameter, not longitudinally.)
Jianyu’s group found this behavior by following the progression of the lithium ions as they travel along the nanowire and create what researchers christened the “Medusa front” — an area where the high density of mobile dislocations causes the nanowire to bend and wiggle as the front progresses. The web of dislocations is caused by lithium penetration of the crystalline lattice.
“These observations also prove that nanowires can sustain large stress — greater than 10 GPa [gigapascals] — induced by lithiation without breaking, indicating that nanowires are very good candidates for battery electrodes,” says Jianyu.
Still, the researchers were surprised to see the lengthwise elongations and the dislocations. Says Jianyu, “No one had ever seen either before. But our observations tell battery researchers how they are generated, how they evolve during charging, and offer guidance in how to mitigate them. This is the closest view to what’s happening during charging of a battery that researchers have achieved so far.”
Lithiation-induced volume expansion, plasticity, and pulverization of electrode materials are the major mechanical defects that plague the performance and lifetime of high-capacity anodes in lithium-ion batteries, Jianyu says. “So our observations of structural kinetics and amorphization [the change from normal crystalline structure] have important implications for high-energy battery design and in mitigating battery failure.”
The electronic noise level generated from the researchers’ measurement system was too high to read electrical currents, but co-author John Sullivan (1132) estimated a current level of a picoampere flowing in the nanowire during charging and discharging. The nanowire was charged to a potential of about 3.5 volts, Jianyu says. A picoampere is a millionth of a microampere. A microampere is a millionth of an ampere.
Functioning in a vacuum environment
The reason that atomic-scale examination of the charging and discharging process of a single nanowire had not been possible was because the high vacuum in a TEM made it difficult to use a liquid electrolyte. Part of the Huang group’s achievement was to demonstrate that a low-vapor-pressure ionic liquid — essentially, molten salt — could function in the vacuum environment.
Although the work was carried out using tin oxide (SnO2) nanowires, the experiments can be extended to other materials systems, either for cathode or anode studies, Jianyu says. “Our experiments lay a foundation for in-situ studies of electrochemical reactions, and will have broad impact in energy storage, corrosion, electrodeposition, and general chemical synthesis research field as well,” he predicts.
Other researchers contributing to this work include Xiao Hua Liu, Nicholas Hudak, Arunkumar Subramanian (all 1132), and Hongyou Fan (1813); Li Zhong, Scott Mao, and Li Qiang Zhang of the University of Pittsburgh; Chong Min Wang and Wu Xu of Pacific Northwest National Laboratory; and Liang Qi, Akihiro Kushima, and Ju Li of the University of Pennsylvania. Funding came from Sandia’s Laboratory Directed Research and Development Office and DOE’s Office of Science through CINT and the Energy Frontier Research Centers program.